COVID-19 Vaccine is Welcome News for Home-Based Palliative Care Clinicians
COVID-19 Vaccine is Welcome News for Home-Based Palliative Care Clinicians


The FDA approval of COVID-19 vaccines is welcomed and offers home-based palliative care clinicians much-needed hope during a challenging time. These hard-working professionals have dealt with limited in-home visits for seriously ill patients while working to prevent hospitalizations, support advance care planning, provide relief to caregivers and ensure that patients have food, medications and social contact — while keeping themselves and their own families safe.
Throughout the pandemic, Turn-Key Health, a CareCentrix company, has amped up its virtual visits to provide clinical and non-clinical home-based palliative care. We have also continued to conduct in-person home-based care to ensure ongoing, coordinated care and assessments.
Frontline Workers in the Home
Vaccination administration began in early January after the F.D.A.’s emergency authorization of the Pfizer-BioNTech vaccine. Trucks and cargo planes packed with the first of nearly three million doses of coronavirus vaccine fanned out across the country, as hospitals in all 50 states set up injection sites. Most of the first injections were given to high-risk health care workers and vulnerable people residing in nursing homes and assisted living centers.
We look forward to when vaccine supplies are more readily available for all adults across the U.S. The COVID-19 vaccine is a welcome relief for our clinicians and those they care for.
Related Blog Posts

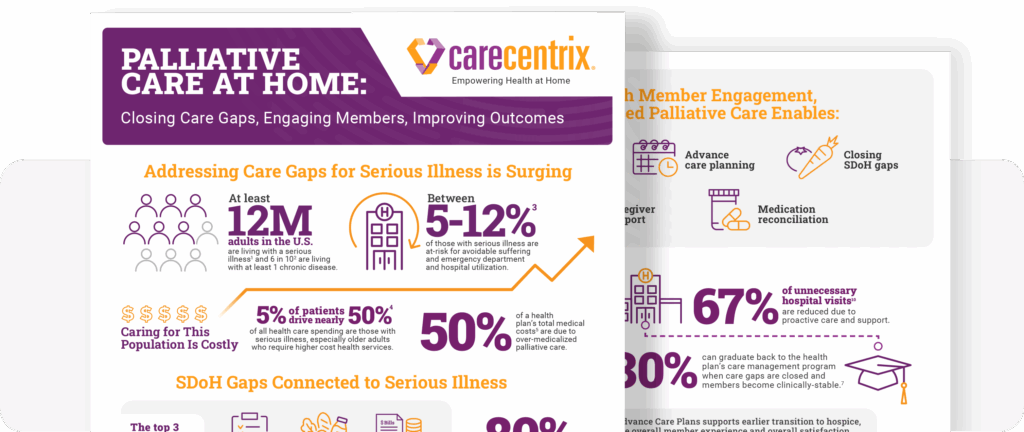
Palliative Care at Home: Closing Gaps, Engaging Members
Supporting members with serious illness can become costly. The need…
Family Caregivers: How Palliative Care Can Help
Facing a serious illness is overwhelming for patients, their family…

Moving Palliative Care from the Hospital to Home
Payors are constantly looking for ways to enhance the member…

Defining Vulnerable Populations in Healthcare
Defining Vulnerable Populations in Healthcare As providers, health plans, and…

4 Healthcare Trends that MA Plans Should Know in 2023
This year will be one marked by significant change and…

The Caregiving Crisis: How Health Plans Can Help
Over the course of my career caring for and developing…
